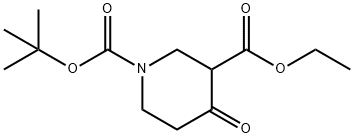
N-Boc-3-carboethoxy-4-piperidone synthesis
- Product Name:N-Boc-3-carboethoxy-4-piperidone
- CAS Number:98977-34-5
- Molecular formula:C13H21NO5
- Molecular Weight:271.31

24424-99-5
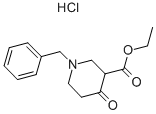
1454-53-1

98977-34-5
Using ethyl 1-benzyl-4-oxo-3-piperidinecarboxylate hydrochloride (10.0 g, 33.6 mmol) and ethanol (800 mL) as the reaction system, a Pd/C catalyst (1.0 g, 10% w/w) was added and the hydrogenation reaction was carried out for about 15 h at room temperature. Upon completion of the reaction, triethylamine (19 mL, 134.3 mmol) and di-tert-butyl dicarbonate (8.0 g, 36.9 mmol) were added to the system, and stirring was continued at room temperature for 16 hours. The reaction mixture was filtered through diatomaceous earth to remove the catalyst and the filtrate was concentrated under vacuum and the residue was dissolved in dichloromethane and washed with water for extraction. The organic phase was separated, dried with anhydrous sodium sulfate and concentrated again in vacuum to give the final ethyl 1-N-Boc-4-oxo-3-piperidinecarboxylate (Compound IB, 9.11 g, 100% yield).

24424-99-5
865 suppliers
$13.50/25G

1454-53-1
240 suppliers
$5.00/5g

98977-34-5
200 suppliers
$5.00/250mg
Yield:98977-34-5 100%
Reaction Conditions:
Stage #1: ethyl 1-benzyl-4-oxopiperidine-3-carboxylate hydrochloridewith hydrogen;palladium 10% on activated carbon in ethanol at 20; under 760.051 Torr; for 15 h;
Stage #2: di-tert-butyl dicarbonatewith triethylamine in ethanol at 20; for 16 h;
Steps:
1; 1.1
A solution of l-benzyl-3-carboethoxy-4-piperidone hydrochloride (IA) (10.0 g, 33.6 mmol) in ethanol (800 mL) and Pd/C (1.0 g, 10% w/w) was hydrogenated at 1 arm. for about 15 hours at room temperature. After the reaction was complete, triethylamine (19 mL, 134.3 mmol) and (Boc)2O (8.0 g, 36.9 mmol) were added to the mixture. The resulting solution was stirred at room temperature for 16 hours and the catalyst was removed by filtering through Celite. The filtrate was concentrated in vacuo, the resulting residue was dissolved in dichloromethane and extracted by washing with water. The organic phase was separated and dried over Na2SO4 and concentrated in vacuo to provide compound IB (9. Ig, 100%).
References:
WO2008/130584,2008,A1 Location in patent:Page/Page column 131; 135

24424-99-5
865 suppliers
$13.50/25G
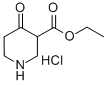
4644-61-5
167 suppliers
$10.00/1g

98977-34-5
200 suppliers
$5.00/250mg

67848-59-3
51 suppliers
$110.00/50mg

24424-99-5
865 suppliers
$13.50/25G

98977-34-5
200 suppliers
$5.00/250mg

24424-99-5
865 suppliers
$13.50/25G

52763-21-0
235 suppliers
$6.00/1g

98977-34-5
200 suppliers
$5.00/250mg
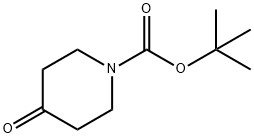
79099-07-3
0 suppliers
$5.00/5g
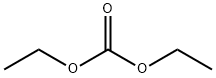
105-58-8
477 suppliers
$15.00/25g

98977-34-5
200 suppliers
$5.00/250mg