| | 134098-61-6 Basic information More.. |
| Product Name: | Fenpyroximate | | Synonyms: | 4-[[[[(1,3-Dimethyl-5-phenoxy-1H-pyrazol-4-yl)methylene]amino]oxy]methyl]-benzoic acid 1,1-dimethylethyl ester;tert-butyl (e)-4-(((((1,3-dimethyl-5-phenoxy-1h-pyrazol-4-yl)methylene)amino)oxy)methyl)benzoate;PAMANRIN;ORTUS;1,1-dimethylethylester,(e)-)methyl);4-(((((1,3-dimethyl-5-phenoxy-1h-pyrazol-4-yl)methylene)amino)oxy)methyl)-,1,1-dimethylethylester,benzoicaci;benzoicacid,4-(((((1,3-dimethyl-5-phenoxy-1h-pyrazol-4-yl)methylene)amino)oxy;nni850 | | CAS: | 134098-61-6 | | MF: | C24H27N3O4 | | MW: | 421.49 | | EINECS: | | | Mol File: | 134098-61-6.mol | 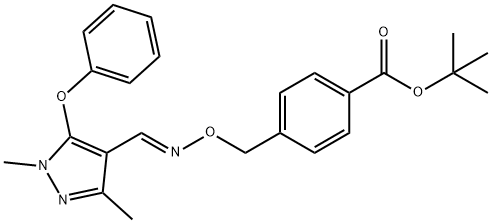 |
Use
When fenpyroximate is administered orally to rats,
radiocarbons from 14C-fenpyroximate are rapidly and
almost completely excreted in the urine and feces
within 72 h, and fenyproximate seems to be
metabolized via oxidation of the tert-butyl group and
methyl group at the 3-position in the pyrazole ring, p-
hydroxylation in the phenoxy moiety, N-demethylation,
hydrolysis of the tert-butyl ester, cleavage of the oxime ether bond, and/or E/Z isomerization. In soils, 12
degradation products are identified, and in sterilized
soils the degradation of fenpyroximate and CO2,
evolution are negligible. Fenpyroximate degrades
through hydrolysis of tert-butyl ester, isomerization or
cleavage of the oxime ether, N-demethylation,
oxidation of the methyl group at the 3-position on the
pyrazole ring, and hydroxylation of the phenoxy ring,
and is finally mineralized to CO2 and/or bound to soil
organic matter.
- Fenpyroximate
-

- US $50.00 / kg
- 2025-01-03
- CAS:134098-61-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000kg
- Fenpyroximate
-

- US $50.00 / kg
- 2025-01-03
- CAS:134098-61-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000kg
- Fenpyroximate
-

- US $50.00 / kg
- 2025-01-03
- CAS:134098-61-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000kg
|
|
134098-61-6
Recommend Suppliers |
|